Liang W.Q., Xie M.W. and Tan Q.G.. 2021. Water Research, 188:116548.
In aquatic environments, the ecological risks posed by metals are greatly affected by water chemistry, thereby creating challenges for water quality management. Biotic ligand models (BLMs) have become the most widely used tools to interpret and predict water chemistry effects. Traditional BLM development methods require a large number of toxicity tests and organisms, and model predictions are limited to certain toxicity statistics (e.g., 48-h median effective concentration, 48-h EC50), to which the models were calibrated. To address these limitations, we propose a new method to develop BLMs by integrating them into the toxicokinetic-toxicodynamic (TK-TD) framework. Metal bioaccumulation was predicted from metal exposure and water chemistry using the BLM-type toxicokinetics, whilst metal toxicity was predicted from metal bioaccumulation using the toxicodynamics. Using the new method, we developed a kinetic BLM of cadmium for Daphnia magna with only six toxicity tests and 1540 daphnids; this represents a 60-80% reduction compared to the traditional methods. The model was validated in the presence of commercial dissolved organic matter (DOM) and in natural waters sampled from 12 lakes. The kinetic BLM was able to accurately simulate the protective effects of the commercial DOM by employing the Stockholm humic model, whilst the complexation capabilities of some natural DOM were overestimated. We further used the model to predict Cd EC50 and no-effect concentrations for different waters, generating predictions close to the effect concentrations reported in the literature. Overall, our method requires fewer resources and presents an easier approach to develop BLMs; the kinetic BLM is more flexible and can serve as a useful tool for developing water quality criteria. (C) 2020 Elsevier Ltd. All rights reserved.
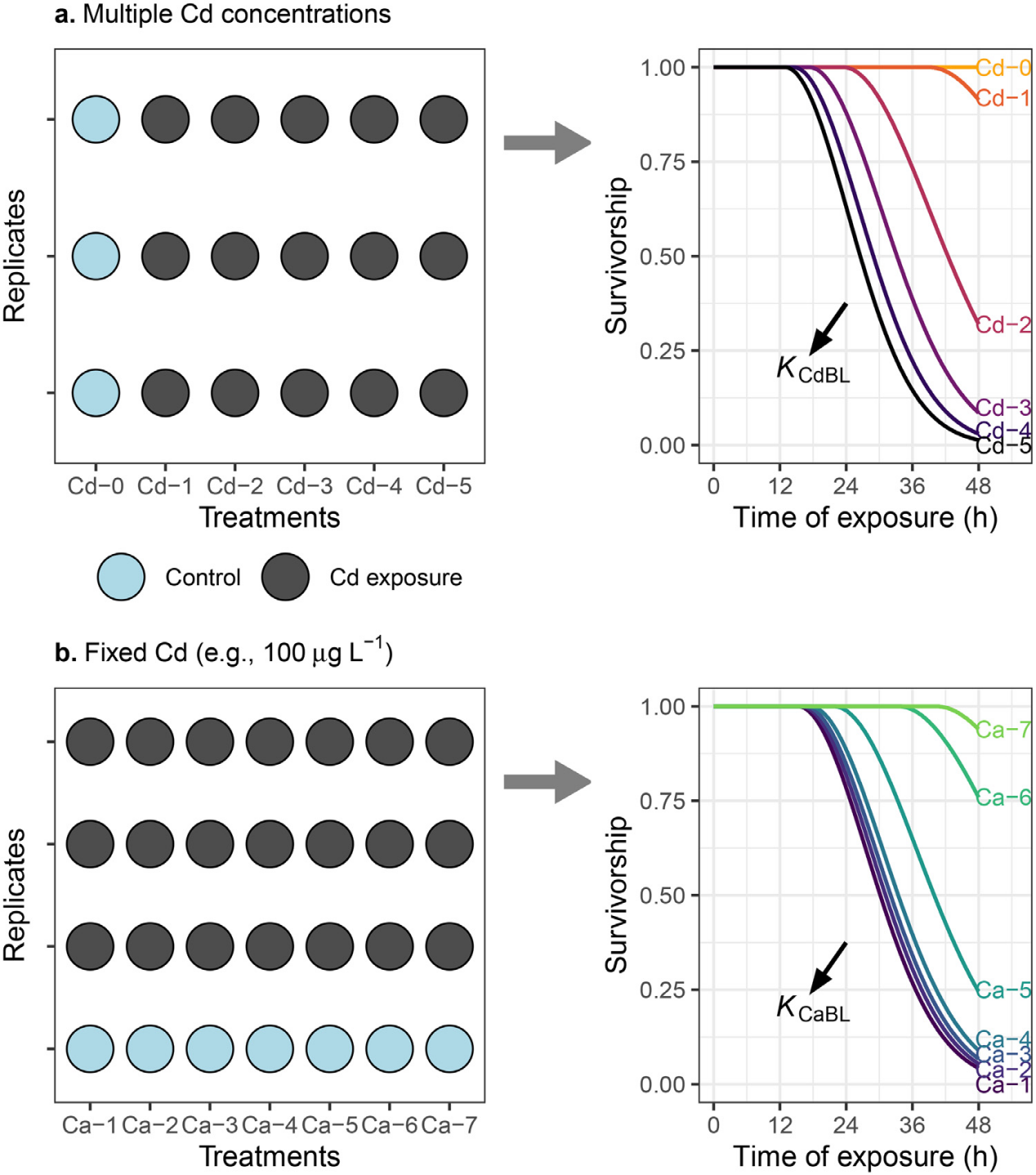
Figure 1. Experimental design for estimating BLM parameter (log K) values. (a) log KCdBL estimated from the Cd-concentration-gradient experiment (Cd-0 to Cd-5: increasing Cd concentration); (b) log KCiBLestimated from the cation competition experiments (using Ca as an example; Ca-1 to Ca-7: increasing Ca concentration). The log K values were estimated by fitting predicted survivorship to the observed survivorship. Control: test solution without adding Cd; Cd exposure: 25−400 μg Cd L−1 in (a) and 100 μg Cd L−1 in (b).

