Zhiteng Chen, Jingqun
Ao, Wenchuan Yang, Liping Jiao, Tianling Zheng, Xinhua Chen,Appl
Microbiol Biotechnol, 2013. 97:10381-10390
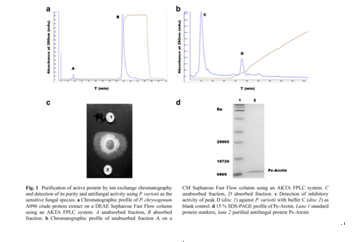
A fungal strain,Penicillium chrysogenumA096,was isolated from an Arctic sediment
sample. Its culture supernatant inhibited mycelial growth of some plant
pathogenicfungi. After saturation ofP.
chrysogenumA096 culture supernatant with ammonium sulfate and ion exchange
chromatography, a novel antifungal protein (Pc-Arctin) was purified
andidentified by matrix assisted laser desorption ionization-time offlight-time
of flight-mass spectrometry (MALDI-TOF-TOF-MS). The gene encoding for Pc-Arctin
consisting of 195 nucleotides was cloned from P. chrysogenum A096 to confirm
themass spectrometry result. Pc-Arctin displays antifungal activityagainstPaecilomyces variotii,Alternaria longipes, andTrichoderma virideat minimum inhibitory
concentrations(MIC) of 24, 48, and 192 ng/disc, respectively. Pc-Arctin wasmost
sensitive to proteinase K and then to trypsin but insensitive to papain.
Pc-Arctin possesses high thermostability andcannot be antagonized by common
surfactants, except forsodium dodecyl sulfate (SDS). Divalent ions, such as Mn2+,Mg2+,
and Zn2+, inhibited the antifungal activity of
Pc-Arctin.Hemagglutination assays showed that Pc-Arctin had nohemagglutinating
or hemolytic activity against red blood cells(RBC) from rabbits, rats, and
guinea pigs. Therefore, Pc-Arctinfrom ArcticP. chrysogenummay represent a novel antifungalprotein with
potential for application in controlling plant pathogenic fungal infection.
返回

